|
With the right integrations, EDCs can receive data from apps and medical devices, Electronic Health Records (EHRs) at the hospitals, and ePRO questionnaires. Today, clinical studies mostly use EDCs to collect CRFs, but modern solutions can go way beyond that. So basically, EDC is a CDMS, and the terms may be used interchangeably Clinical Data Managementis the practice of delivering the best data quality with the least number of mistakes. 
Today, EDC has replaced Clinical Data Management Systems (CDMS) - another acronym that refers to the process rather than to any specific tool. EDC systems are the most widely used eClinical tools with the top solutions including Oracle Health Sciences InForm, Medidata Rave EDC, Bioclinica EDC, and many more.ĮDCs can be as complex or as simple as the study team dictates, but in its core, EDC consists of a graphical user interface, a query management module, and a reporting tool. EDCs have CDISC-compliant templates making it easy to design and export eCRFs. Source: CDISC Electronic Data Capture is a tool for recording and collecting clinical documents. Today, a lot of case report forms are electronic (eCRFs) and they are created and filled using Electronic Data Capture (EDC) software.ĬDASH-compliant eCRF created in Oracle Health Sciences InForm It’s compiled by a group of experts, and in the US, EU, and Japan, a clinical trial protocol should adhere to the Good Clinical Practice guidance - an international ethical and scientific standard for the design and conduct of clinical trials.Ī case report form is a questionnaire that documents the data about each participant and can include as little as information about their current physical condition and as much as data spanning several weeks or months. There are two main documents compiled during trial design - protocols and case report forms (CRFs).Ī protocol describes the objective, methodology, evaluation criteria, and even trial location. Study design and patient data capture using EDC There are four main types of objectives in clinical trials:Ī simplified visualization of main clinical trial systems and their functions At every subsequent stage, dosage is adjusted and the number of participants is increased. It starts with lab testing, followed by human testing. 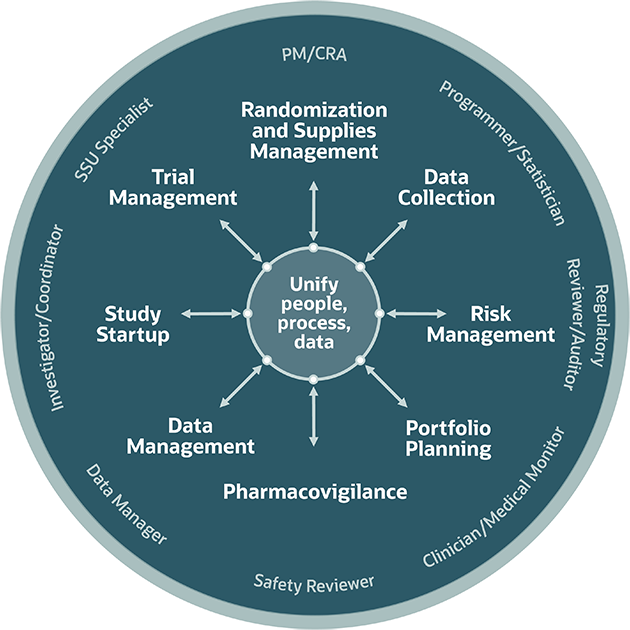 
They often employ a contract research organization (CRO) to plan and conduct the research on behalf of the sponsor. A pharmaceutical, biotechnical, or medical device company sponsors the clinical trial to get their medication or device approved by FDA or an international regulatory authority. What is a clinical trial? Clinical trial workflow and toolkitĪ clinical trial is an experiment done to ensure the safety and efficacy of new treatments. We’ll disclose how these issues can be resolved. Spoiler alert: There are a few areas ripe for improvement. 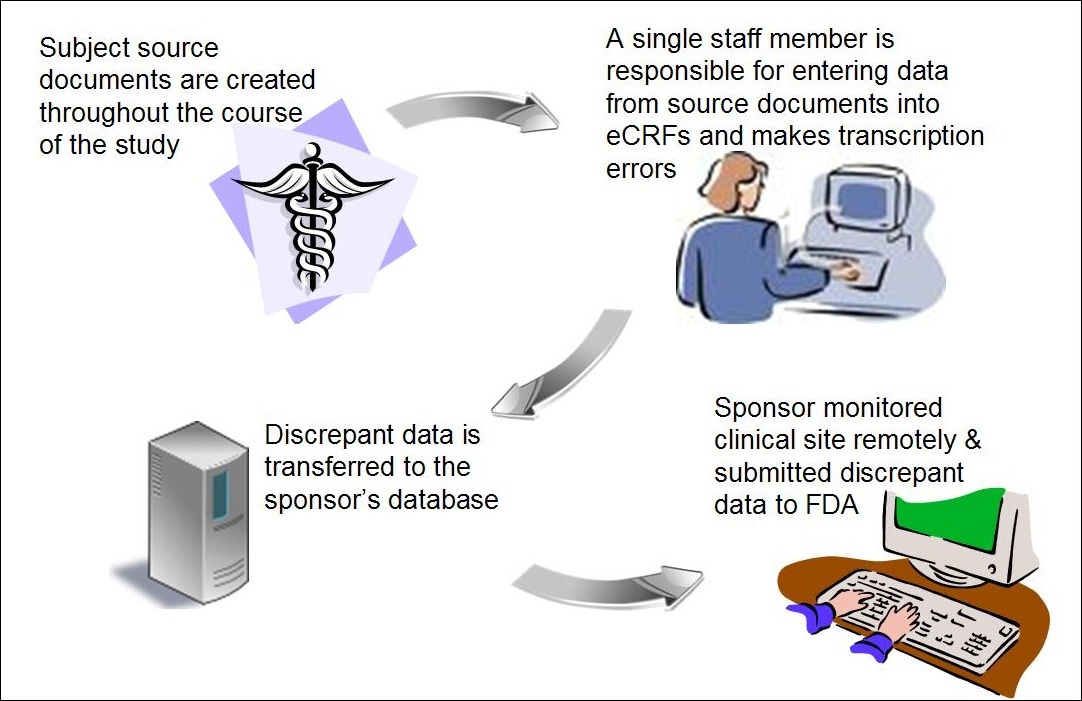
Today we will talk about how clinical trials are done today from the technology point of view. Either way, trial transparency is a big deal, and it’s especially important in times when global collaboration and connectivity is crucial for our wellbeing. Why do we need this registry? Well, WHO believes that the registration of clinical trials is a “scientific, ethical and moral responsibility.” It also avoids duplication and recruits more participants. To be registered, a trial must provide a set of internationally-agreed-upon information about its design, conduct, and administration. This portal, established in 2004, stores the information about trials from all around the world and makes them available to everyone online from a single point of access. At the time of writing this article, 2,898 clinical studies related to COVID-19 are registered on WHO’s International Clinical Trials Registry Platform.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
