
Since the mass number remains unchanged, the daughter nuclide will be germanium-72. 
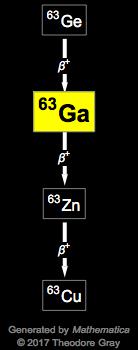
Thallium has been used for identifying thyroid metastases, and gallium 67 is taken up by inflammatory cells, particularly mast cells, and therefore can be used in some cases of destructive thyroiditis such as subacute thyroiditis (Table II). This will be the identity of the daughter nuclide. Iodine-123 decays by electron capture, with a photon energy of 159 keV and a half-life of 13 hours. Germanium, #"Ge"#, has an atomic number of #32#. Look at the element that comes immediately after gallium in the periodic table. You'll find gallium located in period 3, group 13. Grab a periodic table and look for the atomic number of gallium, #"Ga"#. So, you know that gallium-72 has a mass number equal to #72#. On the other hand, its mass number, which represents the number of protons and neutrons located inside the nucleus, will remain unchanged. Such a vacancy can be created by photoionization, ion-atom collisions, electron bombardment, electron capture (EC), or internal conversion electron (CE). #color(purple)(|bar(ul(color(white)(a/a)color(black)(""_ 0^1"n" -> ""_ 1^1"p" + ""_ "-1"^0"e" + bar(nu)_"e")color(white)(a/a)|)))#Īs a result, the atomic number of the daughter nuclide will increase by #1#, since its nucleus will contain an extra proton. This happens because a neutron located inside the nucleus is converted to a proton. The 68 Ge decays to 68 Ga by electron capture with a half-life. 67 Ga is produced at a cyclotron or accelerator by proton irradiation. 67 Ga has a 3.26-day half-life and decays by both electron capture and positron emission.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
